Development of a Soluble Guanylate Cyclase Radioligand Binding Assay Using [3H]-Praliciguat
9th International Conference on cGMP (2019)
This preclinical study provides insights into praliciguat’s mechanism of action by characterizing the nature of its high affinity binding to sGC.
Demonstration of praliciguat synergy with nitric oxide in an sGC activity assay
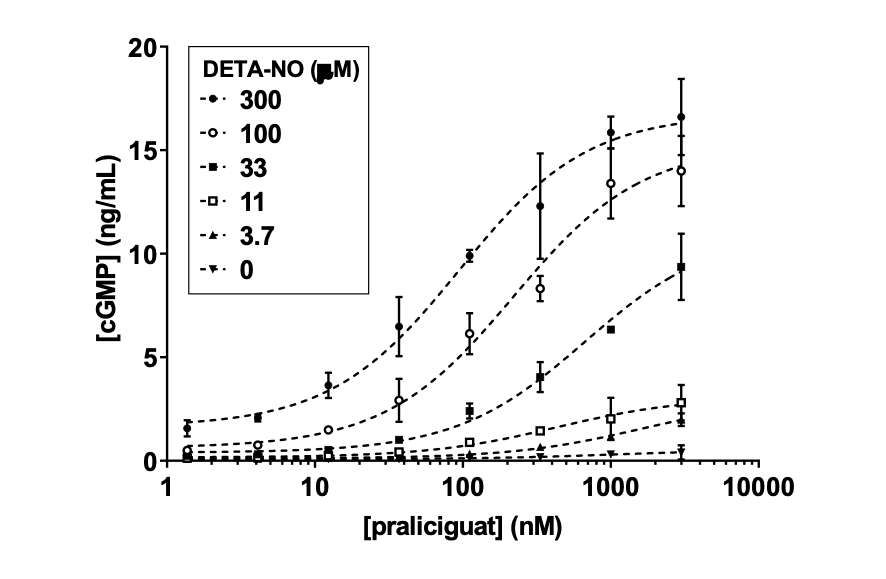
Cyclerion developed a binding assay using [3H]-praliciguat radioligand and size-exclusion chromatography to analyze the binding of praliciguat to purified human recombinant sGC. Kessler et al. (cGMP 2019) poster presentation by Daniel Zimmer
A binding assay to evaluate sGC stimulators
Deborah Dodge, senior scientist at Cyclerion, explains an assay we developed to analyze the binding of Cyclerion’s sGC stimulator praliciguat to sGC. The assay showed that praliciguat binds to sGC with high affinity and requires the presence of nitric oxide, reinforcing our understanding about how praliciguat works. The assay may also further the field of sGC research as it allows scientists to better understand the binding properties of different sGC stimulators.

 Return to Archive
Return to Archive